There was evidence of the interference pattern between both waves, which showed that light could behave as both a particle and a wave.Ī century later, Albert Einsten reinforced the idea through the principles of quantum mechanics.
The output of the light beams was directed by a mirror.īoth beams of light hit a wall in a dark room. The design of the experiment was made so that the beam of light was wider than the card, thus, when placing the card horizontally, the beam was divided into two approximately equal parts. This division is achieved through the use of a 0.2 millimeter card, located parallel to the beam. The English scientist Thomas Young laid the foundations of Schrödinger's atomic model when in 1801 he conducted the experiment to test the wave nature of light.ĭuring his experimentation, Young divided the emission of a beam of light that crosses a small hole through an observation chamber. The atomic model of Schrödinger is based on the Broglie hypothesis, and on the previous atomic models of Bohr and Sommerfeld.įor this, Schrödinger relied on Young's experiment, and based on his own observations, developed the mathematical expression that bears his name.įollowing the scientific foundations of this atomic model: Young's experiment: the first demonstration of wave-particle dualityīroglie's hypothesis about the undulatory and corpuscular nature of matter can be demonstrated by the Young Experiment, also known as the double-slit experiment. The model does not contemplate the stability of the nucleus, it only refers to explaining the quantum mechanics associated with the movement of electrons within the atom. These atomic orbitals have different levels and sub-levels of energy, and can be defined between electron clouds. The orbitals describe a movement of translation around the nucleus of the atom. These areas of probability are called atomic orbitals. It only establishes a probability zone to locate the electron.
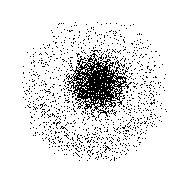
This model does not predict the location of the electron, nor does it describe the route it makes within the atom. The electrons move constantly, that is, they do not have a fixed or defined position within the atom. Describes the movement of electrons as standing waves.

Orbital 1s, 2s and 2p within a sodium atom. 2.1 Young's experiment: the first demonstration of wave-particle dualityĬharacteristics of the Schrödinger atomic model.1 Characteristics of the Schrödinger atomic model.

